Study design and population
This prospective study utilized data from the Trøndelag Health Study (HUNT), Norway’s largest population-based survey26. Details on the HUNT study have been described elsewhere26. Briefly, HUNT encompasses four phases (HUNT1: 1984–1986, HUNT2: 1995–1997, HUNT3: 2006–2008, and HUNT4: 2017–2019), collecting health data through self-reported questionnaires, clinical examinations, and laboratory tests26. In this study, we adhered to the guidelines outlined in the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE)27.
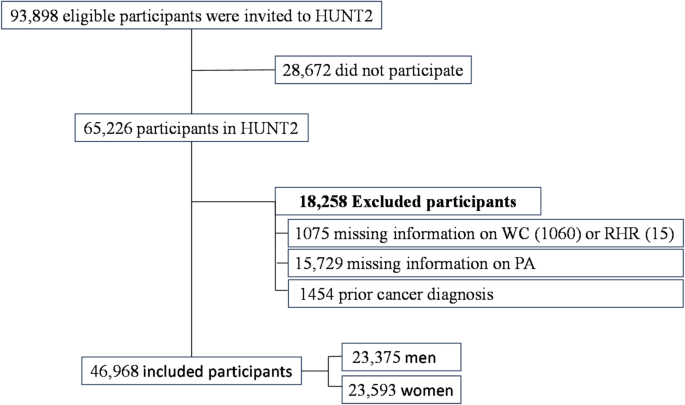
Data from HUNT2 (1995–1997) were used, where 93,898 eligible participants aged ≥ 20 were invited. Out of these, 65,226 (69.5%) individuals participated. First, we excluded 1075 participants without information on WC (n = 1060) or RHR (n = 15). Second, we excluded those who had missing information on PA (n = 15,729). Lastly, participants with prior cancer diagnoses (n = 1454) were excluded. Consequently, the final analysis comprised 46,968 cancer-free participants, including 23,375 (49.8%) men and 23,593 (50.2%) women (Fig. 1).
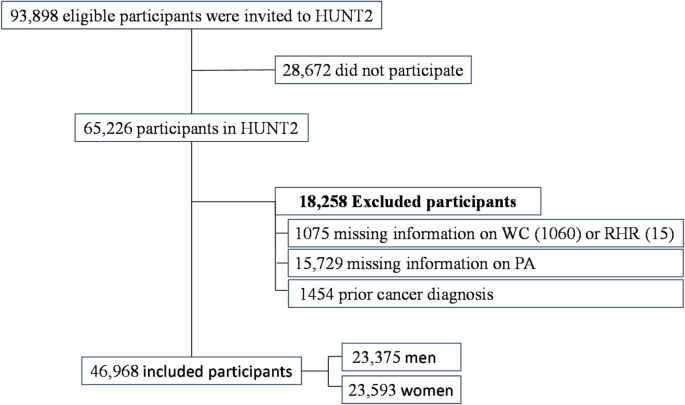
The flowchart illustrates the exclusion criteria for participants in this study. Participants were excluded mainly due to a lack of data on waist circumference (WC), resting heart rate (RHR), or physical activity (PA), or prior cancer diagnosis at baseline.
Estimated CRF
Estimated CRF was calculated from non-exercise prediction models. The models aimed at predicting eCRF in VO2peak (ml/kg/min) and were derived from a sample of healthy middle-aged adults from the HUNT3 population25 and further adapted for use in HUNT221. The variables included in the prediction models were age, WC, RHR, and self-reported PA. The models and the self-reported PA questionnaires were previously validated21,25,28, suggesting high comparability with other non-exercise prediction models25,29,30.
The prediction models used for estimating CRF are as follows25:
For men (Goodness of fit statistic = 0.58, Standard error of estimate = 5.88):
$$105.91 -(0.334 \times \text{Age}) – (0.402 \times \text{WC}) – (0.144 \times \text{RHR}) + (3.102 \times \text{PA}_{\text{ACSM}}\bigr)$$
For women (Goodness of fit statistic = 0.52, Standard error of estimate = 5.37):
$$78.00 – (0.297 \times \text{Age}) – (0.270 \times \text{WC}) – (0.110 \times \text{RHR}) + (2.674 \times \text{PA}_{\text{ACSM}}\bigr)$$
To measure WC, a metal tape was used at the participants’ umbilicus level while standing with the arms flattened and registered to the nearest centimeter18,31. RHR was calculated as the mean of three measurements recorded after sitting for two minutes using Dinamap 845XT (Critikon). Data on PA was obtained from the self-reported questionnaires. Participants answered questions about their intensity levels and duration of leisure-time PA during the past year. Intensity was divided into: light, defined as “not sweating/being out of breath” and vigorous, defined as “sweating/out of breath”. The duration of each intensity level was reported as an average of hours per week (0, < 1 h, 1–2 h, or ≥ 3 h). We initially classified participants’ levels of PA into four categories: inactive (no activity, or ≤ 2 h light activity only), low (≥ 3 h light activity only, or ≤ 2 h light activity and < 1 h vigorous activity), moderate (≥ 3 h light activity and < 1 h vigorous activity, or 1–2 h vigorous activity regardless of light activity), and high (≥ 3 h vigorous activity regardless of light activity)32. This classification was widely used in studies utilizing HUNT2 and showed a dose-response association of PA levels with overall cancer incidence and mortality18,32.
To calculate eCRF, we further classified PA into two categories (PAACSM): a value of “1” was given to those who had moderate or high PA levels. They were regarded as meeting the American College of Sports Medicine (ACSM) recommendations, i.e., at least 75 min (min) per week of vigorous intensity exercise or at least 150 min per week of moderate intensity exercise; and a value of “0” was given to those who were inactive or had low PA level. They were regarded as not meeting the ACSM recommendations21.
Estimated CRF was further classified into age-specific (based on a 10-year interval) quintiles within each sex group to account for the possible curvilinear association between age and CRF and reduce age-related misclassification33. This approach, recommended by Kokkinos et al. 33, helps minimize variability in both mean age and sample size across fitness categories and, therefore, improve the validity of associations with health outcomes. Then, we merged the quintiles into three broader groups: the first quintile as the 20% low eCRF, the second and third quintiles as the 40% medium eCRF, and the fourth and fifth quintiles as the 40% high eCRF group. This reclassification was derived from the Aerobics Center Longitudinal Study (ACLS) and applied here to optimize statistical power34. We have also classified eCRF into age and sex-specific tertiles to be consistent with other previous literature and to assess the robustness of our results from the ACLS classification16,18.
Follow-up and the assessment of the outcome
The HUNT2 data were linked with the Cancer Registry of Norway using the personal identification number. The cancer outcomes were identified using the International Statistical Classification of Disease and Related Health Problems (ICD-10)35. Urinary tract cancers, bladder cancer, and kidney cancer were coded as (C64–C68), (C67), and (C64), respectively. The follow-up period for participants started from baseline until one of the following occurred: first diagnosis of any of these cancers, death, emigration from the county, or the year-end of 2018.
Statistical analysis
Baseline characteristics across the three age-specific eCRF categories were presented separately for men and women using descriptive statistics: means (standard deviations) for continuous variables and counts (percentages) for categorical variables. We used cause-specific Cox proportional hazards (PH) models to assess the relationships between eCRF and the incidence of urinary tract cancers, along with the site-specific incidence of bladder cancer and kidney cancer36,37. By employing cause-specific hazard models, we aimed to evaluate a potentially etiological association of eCRF with the instantaneous rate of cancer occurrence among individuals who remained event-free over time. Crude (age as the timescale) and adjusted hazard ratios (HRs) with 95% confidence intervals (CIs) were calculated in three models separately (Crude model, Model 1, and Model 2), with low eCRF being the reference group. In addition to age in the Crude model, Model 1 adjusted for sex (in total cohort only), sitting time, smoking, alcohol consumption, education, occupational class, family history of cancer, hypertension, and diabetes mellitus. Model 2 was further adjusted for body mass index (BMI) and PA as a part of the sensitivity analysis, as WC and PA were already included in the eCRF Equation20. Details on variable classifications are provided in the Supplementary Methods. Hazard ratios from Model 1 were used as the primary estimates. Analyses were conducted for the total cohort and stratified by sex. To assess trends across eCRF categories, we included eCRF as an ordinal variable in the Cox regression models and reported the corresponding P-values for trend in all the models. We also generated cumulative hazard curves stratified by eCRF categories, based on the cause-specific Cox models, to visually illustrate differences in the rate of event occurrence over time36. Missing data on confounders were handled using the missing-indicator method by assigning a separate “unknown” category in the main analyses.
PH assumptions were tested using Schoenfeld residuals, and there was no evidence of potential violations. To test effect modification by sex in the association between eCRF and the incidence of urinary tract cancers, bladder cancer, or kidney cancer, we compared the Cox PH models with and without including an interaction term between eCRF and sex (eCRF*sex) using the likelihood ratio test (LRT).
Several sensitivity analyses were conducted. First, excluding the first three years of follow-up to address reverse causality. Second, analyzing eCRF tertiles separately to assess the robustness of our results from the ACLS classification. Third, conducting multiple imputations using chained equations (MICE) under the assumption that data were missing at random. Here, we imputed 20 datasets for nine covariates with missing values, including sitting time, smoking, alcohol consumption, education, occupational class, hypertension, diabetes mellitus, BMI, and PA. Fourth, we repeated the analyses using time-varying covariates among participants who took part in both HUNT2 and HUNT3. In this approach, follow-up time was split at the participation date of HUNT3 using the stsplit function in Stata. Information on covariates in Model 1, such as sitting time, smoking, alcohol consumption, family history of cancer, hypertension, and diabetes mellitus was updated from HUNT3, and the other covariates such as education and occupational class were held constant. All analyses were performed using Stata version 18.0 (StataCorp, College Station, TX, USA).
link